Everything's Here sblock Elements and pblock Elements
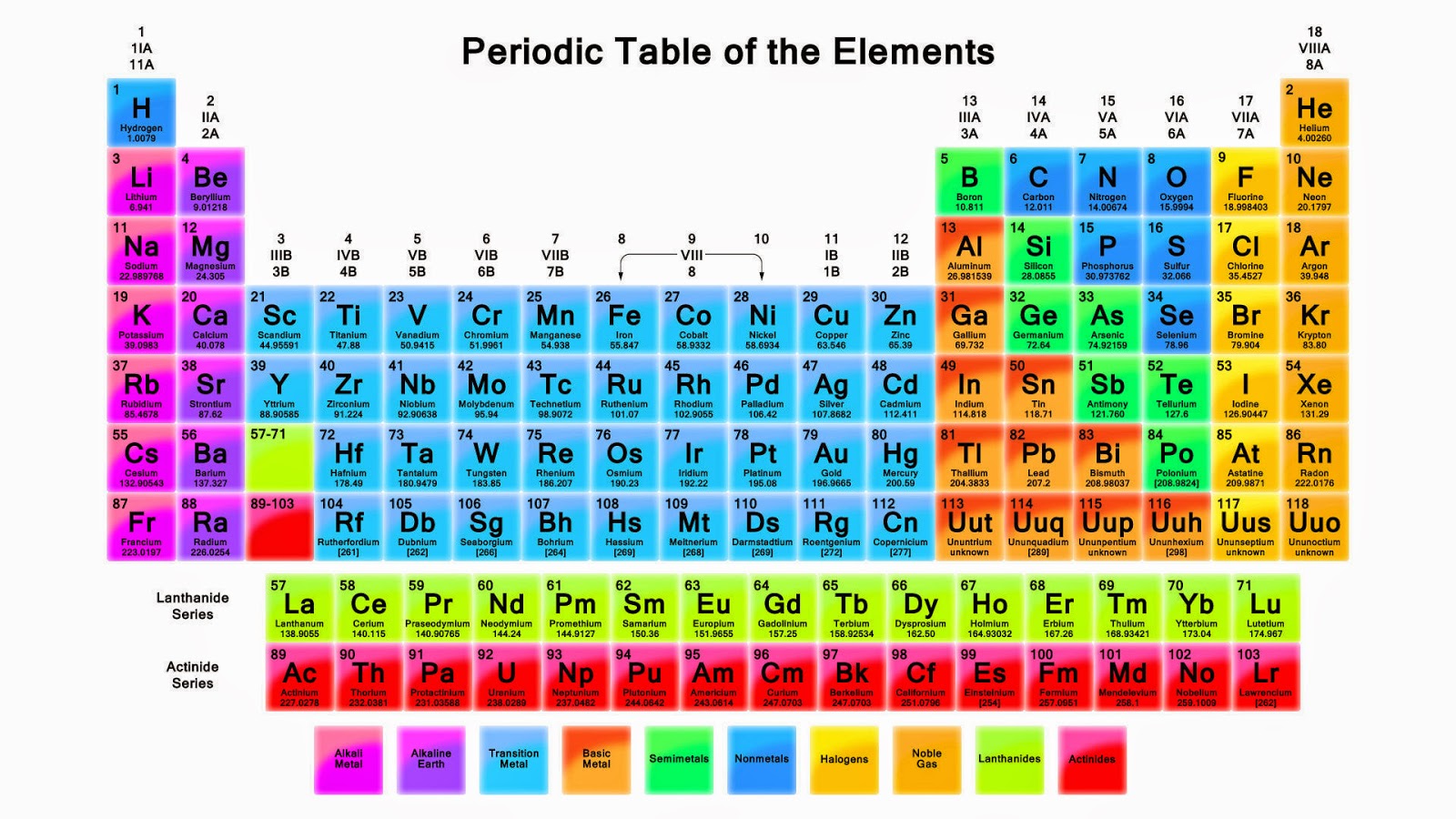
The periodic table is classified into four blocks based on which subshell the valence electron enters. They are namely s,p,d and f blocks. s Block Elements The s block elements are situated at the extreme left side of the periodic table. They include Group 1 elements (Alkali metals), Group 2 elements (Alkaline earth metals), Hydrogen and Helium.
PPT Chapter 5 Electrons In Atoms PowerPoint Presentation, free
P Block Elements Elements in which the final electron enters one of the three p-orbitals of their respective shells are referred to as P block elements. Because a p-subshell has three degenerate p-orbitals, each of which can accommodate two electrons, there are a total of six groups of p-block elements in a p-subshell.
Periodic Table As Per Blocks
The periodic table is divided into four blocks, namely s-block, p-block, d-block, and f-block, which correspond to specific regions of the periodic table where electrons are filled in the s, p, d, and f subshells. The s-block elements have their outermost electrons in the s-subshell, while the p-block elements have their outermost electrons in.

A block diagram of the periodic table shows which sublevels are being
This block contains the elements of groups 3 to 12 of the periodic table. The three series of transition metals are known 3d series, 4d series and 5d series. f-Block Elements The f-block consists of two series lanthanides and actinides of the periodic table. The electronic configuration of actinides is irregular.

Download Periodic Table Of Elements S P D F Blocks Online Printable PDF DOC
Each orbital can be represented by specific blocks on the periodic table. The s-block is the region of the alkali metals including helium (Groups 1 & 2), the d-block are the transition metals (Groups 3 to 12), the p-block are the main group elements from Groups 13 to 18, and the f-block are the lanthanides and actinides series.
Periodic Table Split Into Blocks
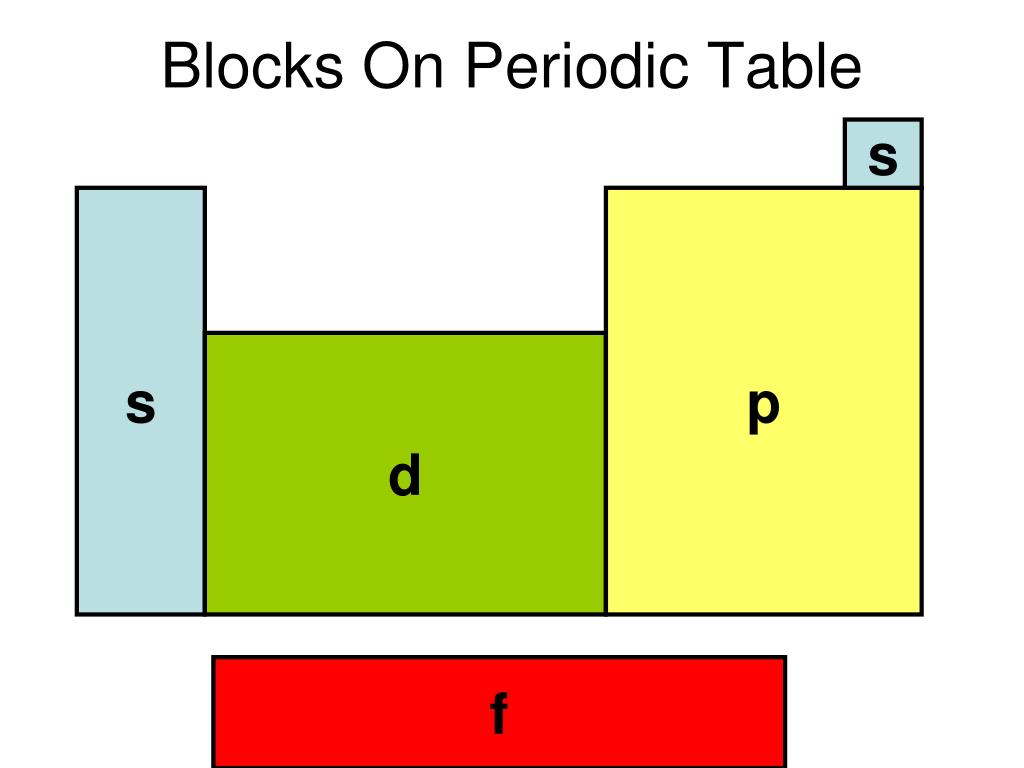
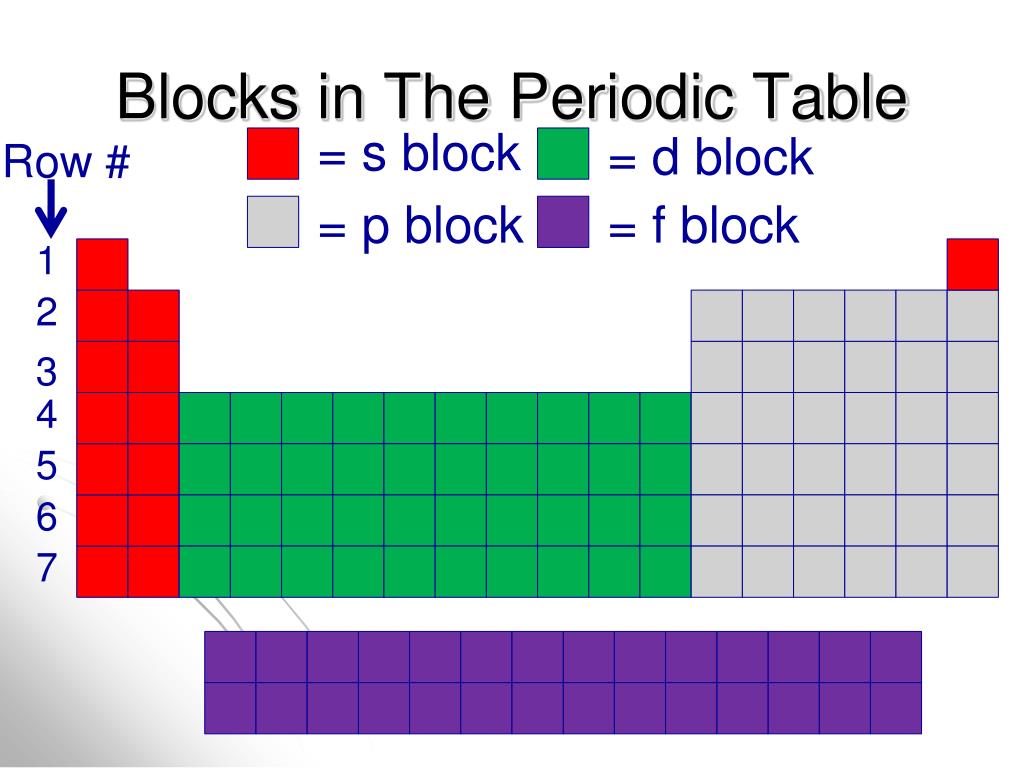
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. Each block is named after its.

Periodic Table Blocks S P D F
The periodic table of s‐, p‐, d‐, and f‐block elements. Source publication +4 The Pivotal Role of s‐, p‐, and f‐Block Metals in Water Electrolysis: Status Quo and Perspectives.
Periodic Table Blocks S P D F Periodic Table Timeline
Steps for Identifying S, P, D, & F -Block Elements. Step 1: Find the element on the periodic table. Step 2: Use periodic table landmarks and mnemonic devices to determine the block.

Periodic Table Blocks S P D F Periodic Table Timeline
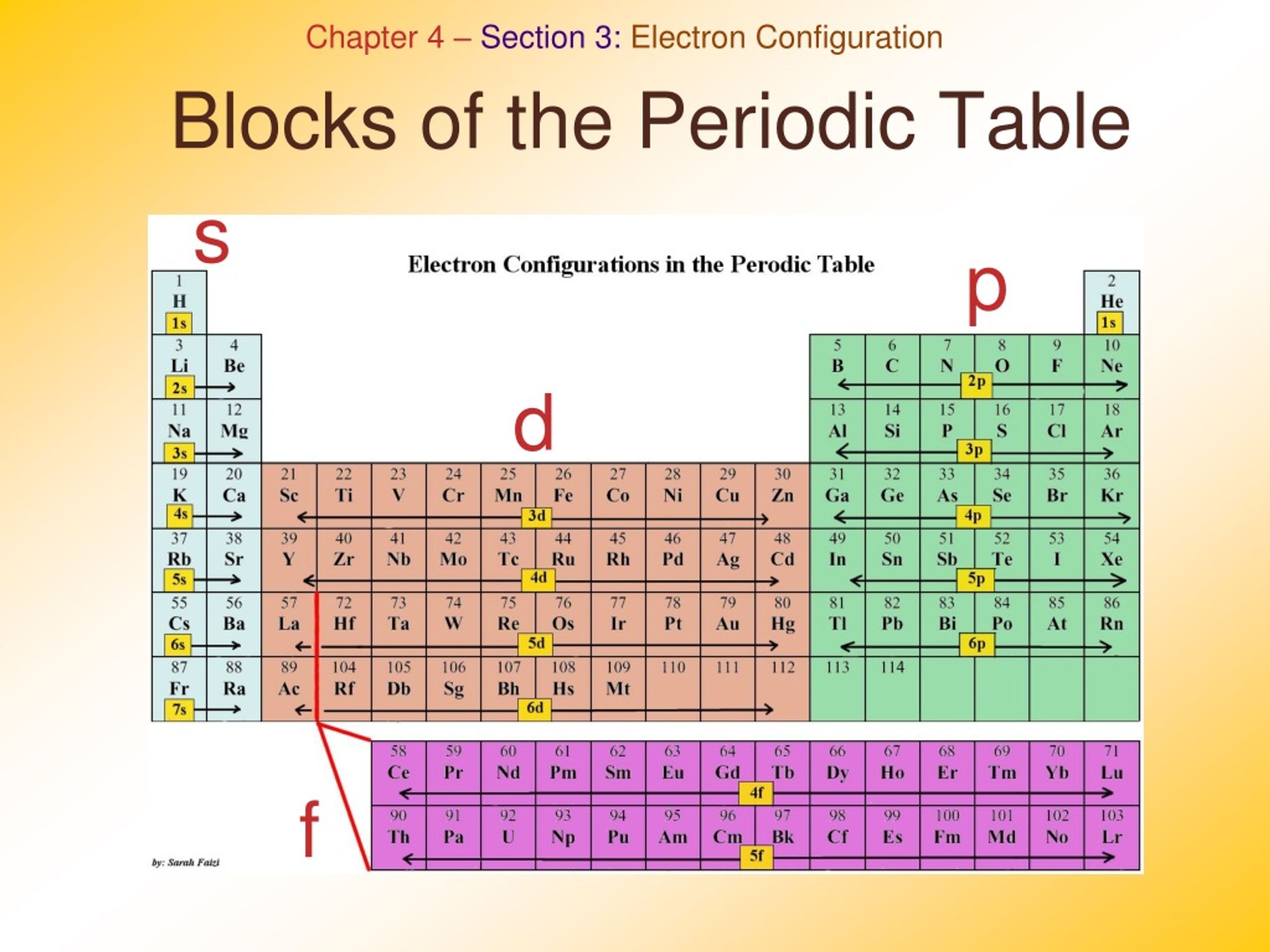
The labels s, p, d and f blocks of the Periodic Table refer to the subshell that is being filled with electrons. ⚛ Group 1 elements occur at the beginning of a new row (Period) of the Periodic Table. The highest energy level (valence shell) contains only 1 electron in an s subshell.

Representative Elements Periodic Table
So while there is a possible number of 32 elements in the period, the current number is slightly less. The period to which a given element belongs can be easily determined by its electron configuration. For example, consider the element nickel (Ni) ( Ni). Its electron configuration is [Ar] 3d8 4s2 [ Ar] 3 d 8 4 s 2.

Periodic Table Blocks S P D F Periodic Table Timeline
A block of the periodic table is a set of groups of chemical elements whose valence electrons occupy, in the ground state, orbitals that share the same azimuthal quantum number ℓ, i.e. belonging to the same sub-electronic layers.
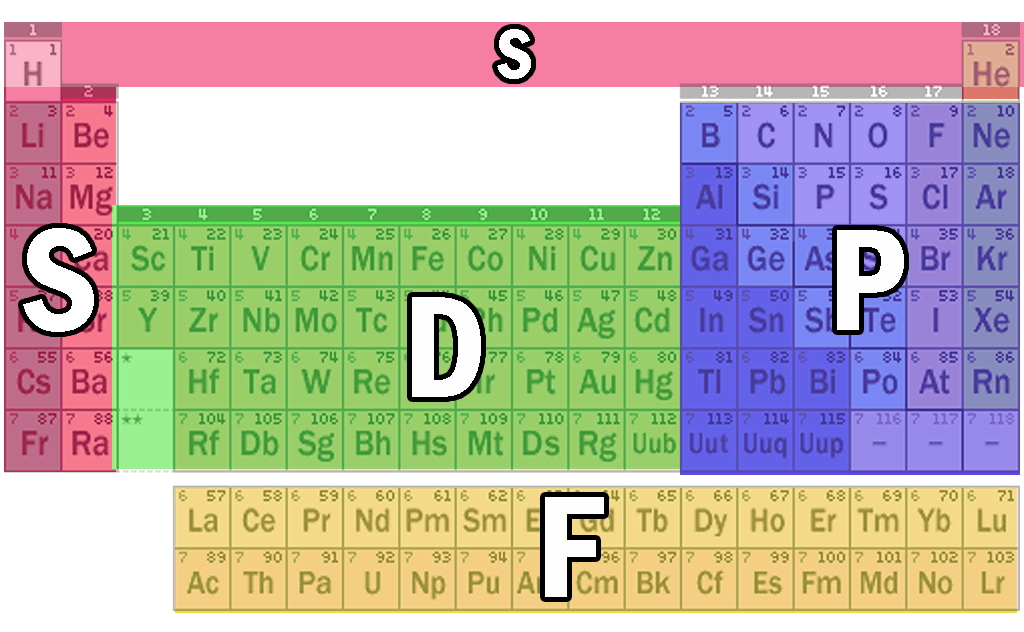
Modern Periodic Table S P D F Blocks Periodic Table Timeline
The completely filled d orbitals count as core, not valence, electrons. The two far-left columns comprise the s -block and the six far-right columns constitute the p -block. The noble gases, which are a part of the p -block, all have eight valence electrons except for helium, which has two. These elements are highly stable and unreactive.
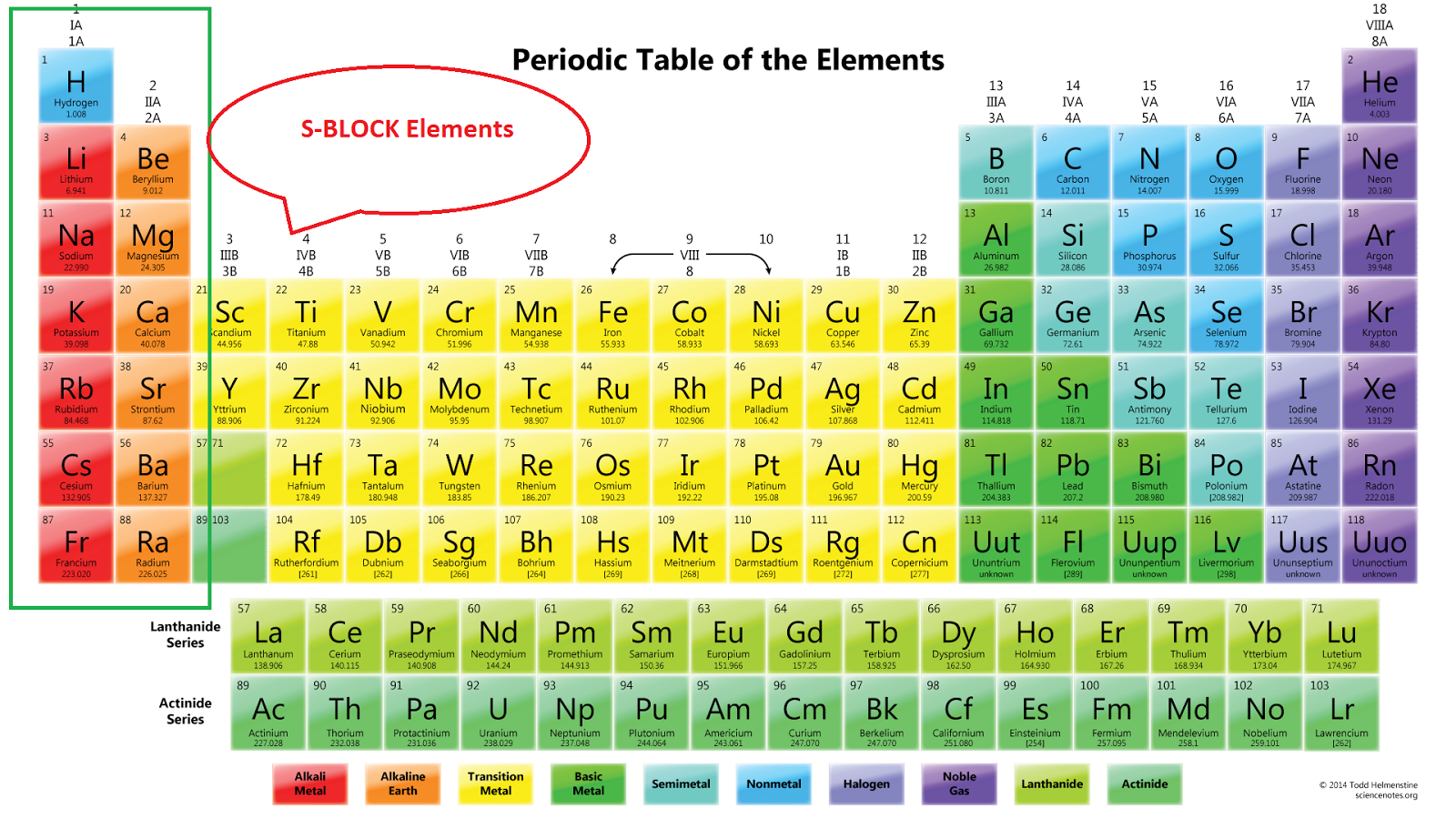
S P D F Block Elements Periodic Table Periodic Table Timeline
These are s, p, d, and f block elements that constitute the whole periodic table. The term block was used by Charles Janet for the first time when he introduced his left step periodic table (LSPT). The divisions into the blocks are characterized by their distinctive nature. For example:

Periodic Table Of Elements With Orbitals
Periodic table history Sets of elements By periodic table structure Groups (1-18) 1 ( alkali metals) 2 (alkaline earth metals) 3 4 5 6 7 8 9 10 11 12 13 14 15 (pnictogens) 16 (chalcogens) 17 (halogens) 18 (noble gases) Periods (1-7,.) 1 2 3 4 5 6 7 8+ Blocks (s, p, d, f,.) Atomic orbitals Aufbau principle
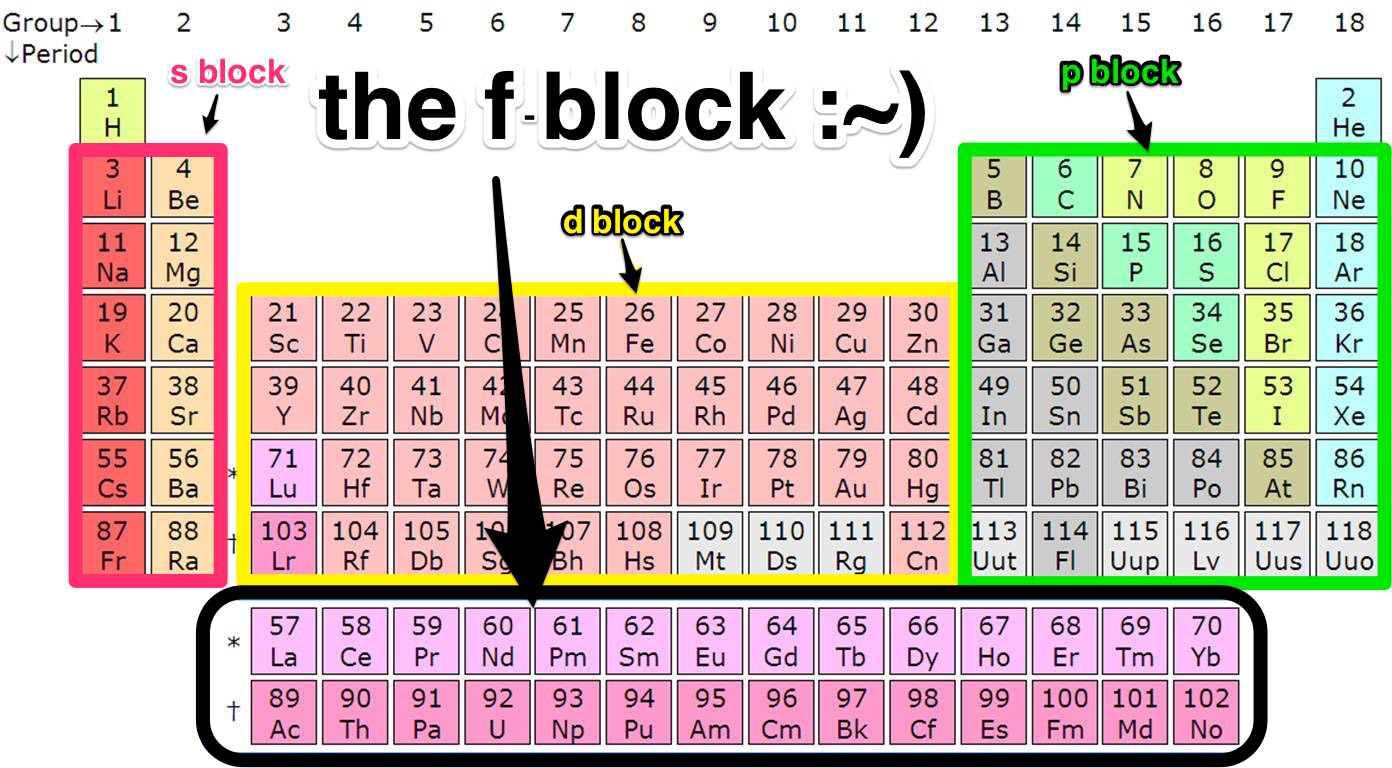
The FBlock An introduction » Scienceline
Watch more of this topic at http://bit.ly/1YxZcRFGET MORE CLUTCH!VISIT our website for more of the help you need: http://bit.ly/1YxZhVcSUBSCRIBE for new vi.
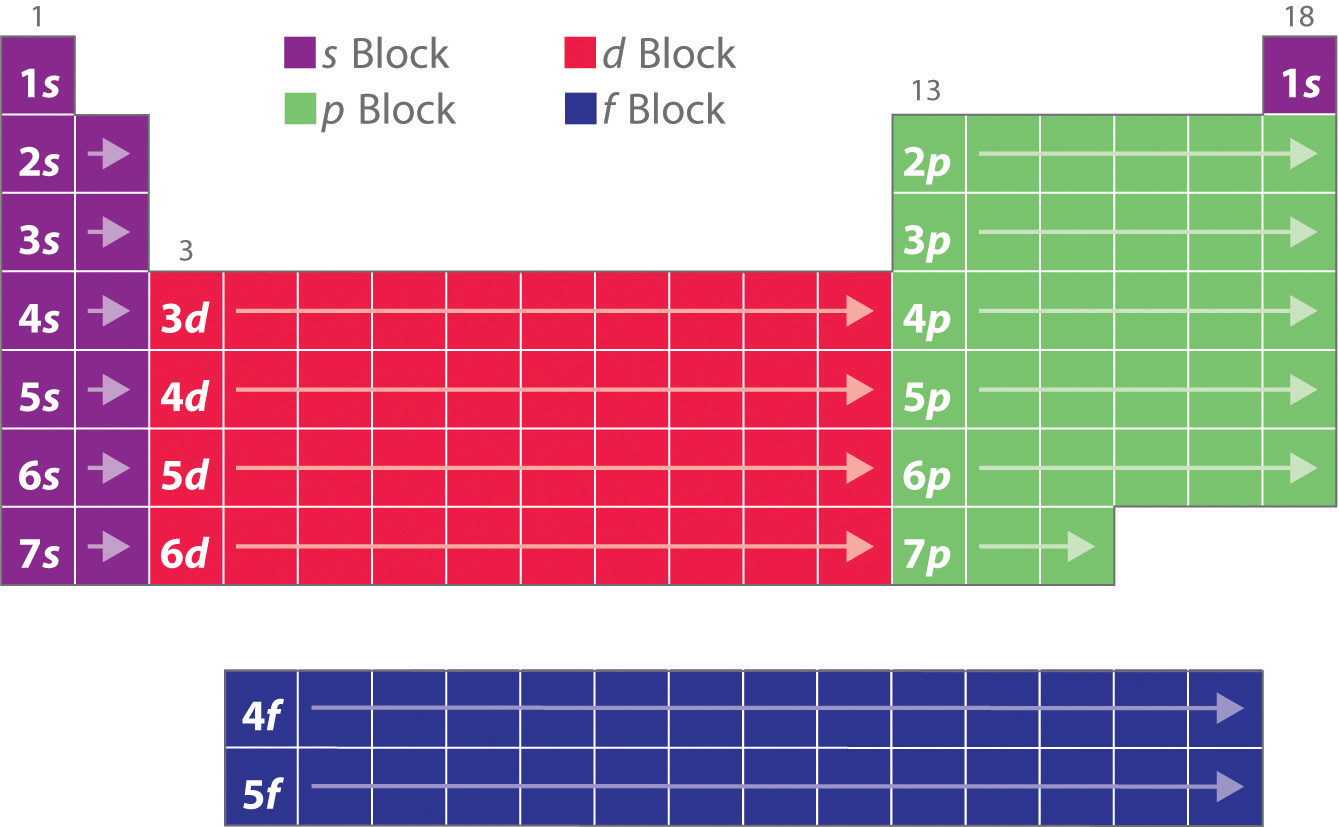
Periodic Table Blocks S P D F Periodic Table Timeline
Here are the features 1) Horizontal rows are called period. They were called series in Mendeleev Periodic Table. There are altogether 7 periods in the Periodic table 2) Vertical columns are called groups. Elements have similar outer electronic configuration are arranged in same groups. There are total 18 groups numbered 1 to 18.